From the patent and other places, I have seen claims of light element waste products in the ash that have been produced during the Rossi reaction.
There are other traces of elements in the used nickel, like Sulfur, Calcium Chlorine, Potassium. Apparently this could be an indication of some fission happening together with the fusion.
Form W-L theory, one can deduce the “secret” catalectic components present in the Rossi process as aluminum.They claim to demonstrate production of lighter elements by the presence of zinc, sulfur, calcium, potassium, silicon, chlorine, copper.
In the periodic table, stating from aluminum and assenting in A order you find as follows:
13- Silicon (mentioned as ash)
14 - Phosphorus
15 – Sulfur (mentioned as ash)
16 – Chlorine (mentioned as ash)
17 – Argon (outgased ?)
18 – Potassium (mentioned as ash)
19 – Calcium (mentioned as ash)
This is consistent with the W-L theory rule for inverse beta decay in the carbon cycle A -> A + 1.

This implies specifically, Raney nickel. Raney nickel is a solid catalyst composed of fine grains of a nickel-aluminium alloy, used in many industrial processes. It was developed in 1926 by American engineer Murray Raney as an alternative catalyst for the hydrogenation of vegetable oils in industrial processes. More recently it is used as a heterogeneous catalyst in a variety of organic syntheses, most commonly for hydrogenation reactions.
Murray Raney graduated as a from the University of Kentucky in 1909. In 1915 he joined the Lookout Oil and Refining Company in Tennessee and was responsible for the installation of a electrolytic cell for the production of hydrogen which was used in the hydrogenation of vegetable oils. During that time the industry used a nickel catalyst prepared with Nickel(II) oxide.
Believing that better catalysts could be produced, around 1921 he started to perform independent research while still working for Lookout Oil. In 1924 a 1:1 Ni/Si alloy was produced, which after treatment with sodium hydroxide, was found to be five times more active than the best catalyst used in the hydrogenation of cottonseed oil
Subsequently, Raney produced a 1:1 Ni/Al alloy following a procedure similar to the one used for the nickel-silicon catalyst. He found that the resulting catalyst was even more active and filed a patent application in 1926. This is the preferred alloy composition for production of Raney nickel catalysts currently in use.
Following the development of Raney nickel, other alloy systems with aluminum were considered, of which the most notable include copper, ruthenium and cobalt
Further research showed that adding a small amount of a third metal to the binary alloy would promote the activity of the catalyst. Some widely used promoters are zinc, molybdenum, and chromium.
Properties
Macroscopically, Raney nickel is a finely-divided gray powder. Microscopically, each particle of this powder is a three-dimensional Mesh consisting of a semi-permeable barrier made of connected strands of metal, fiber, or other flexible/ductile material. This Mesh is similar to web or net in that it has many attached or woven strands with pores of irregular size and shape of which the vast majority are created during the leaching process. Raney nickel is notable for being thermally and structurally stable as well has having a large BET (Brunauer-Emmett-Teller ) surface area.. These properties are a direct result of the activation process and contribute to a relatively high catalytic activity.
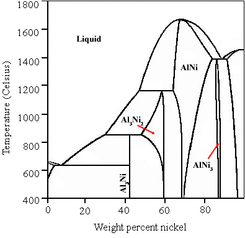
During the metal foaming activation process, aluminum is leached out of the NiAl3 and Ni2Al3 phases that are present in the alloy, while most of the aluminum that remains does so in the form of NiAl. The removal of aluminum from some phases but not others is known as "selective leaching”.
Selective leaching, also called dealloying, demetalification, parting and selective corrosion, is a corrosion type in some solid solution alloys, when in suitable conditions a component of the alloys is preferentially leached from the material.
It has been shown that the NiAl phase provides the structural and thermal stability of the catalyst. As a result, the catalyst is quite resistant to decomposition ("breaking down", commonly known as "aging"). This resistance allows Raney nickel to be stored and reused for an extended period; however, fresh preparations are usually preferred for laboratory use. For this reason, commercial Raney nickel is available in both "active" and "inactive" forms.
The surface area is typically determined via a BET measurement using a gas that will be preferentially adsorbed on metallic surfaces, such as Hydrogen.
It has been shown that almost all the exposed area in a particle of the catalyst has nickel on its surface. Since nickel is the active metal of the catalyst, a large nickel surface area implies that there is a large surface available for reactions to occur simultaneously, which is reflected in an increased catalyst activity. Commercially available Raney nickel has an average nickel surface area of 100 square meters per gram of catalyst.
A high catalytic activity, coupled with the fact that hydrogen is absorbed within the pores of the catalyst during activation, makes Raney nickel a useful catalyst for many hydrogenation reactions. Its structural and thermal stability (i.e., the fact that it does not decompose at high temperatures) allows its use under a wide range of reaction conditions.
Additionally, Solubility of Raney nickel is negligible in most common laboratory solvents, with the exception of Mineral acid such as hydrochloric acid and its relatively high density (about 6.5 g/cm3) also facilitates its separation from a liquid phase after a reaction is completed.