Maybe I can rotate the graph, if that makes it more obvious which way the 'total binding energy' axis goes....
Again, it depends on how you define "fusion". Most people have buried in their definition of "fusion" the assumption that like size particles are involve. D-D, D-T, C-C, 28Si+30Si=58Ni... So "fusion" is the combination of LIKE size smaller particles. To them "fusion" would be Ni+Ni and your implication above holds. Attempting to "fuse" Ni nuclei would be endothermic. But "fusing" a VERY small particle (n, p, D(?)) to ANYTHING (except the A=4,12,16?,20? chain) is still exothermic, no matter WHAT the other particle is. This is because the binding energy (either per nucleon or per nucleus) of the REALLY small particles is so low to start with.D Tibbets wrote: That Ni62 is the peak, tells you that energy can be released as you approach this point from either side- fusion of lighter elements and fission of heavier elements. The claim that fusion will continue to release energy past Ni62 is the bone of contention.
Said statements of the energy balance have NOT been ignored. I have made MULTIPLE references to Ni+Ni being endothermic in complete agreement with your sources. You just seem to be totally unwilling to grasp the point that said statement are about LIKE SIZE PARTICLES. Not about nuclear transmutation in the manner of Ni+p.Then he wrote:I have given astronomical considerations that I think would have obvious and possibly dire consequences if this occurred. I have also included multiple quotes from authoritative sources that state the fusion/ fission energy balance in relation to Ni62 (in some they use Fe56). That these are completely ignored is what is appalling. That my efforts at clarification may be inept is irrelevant.
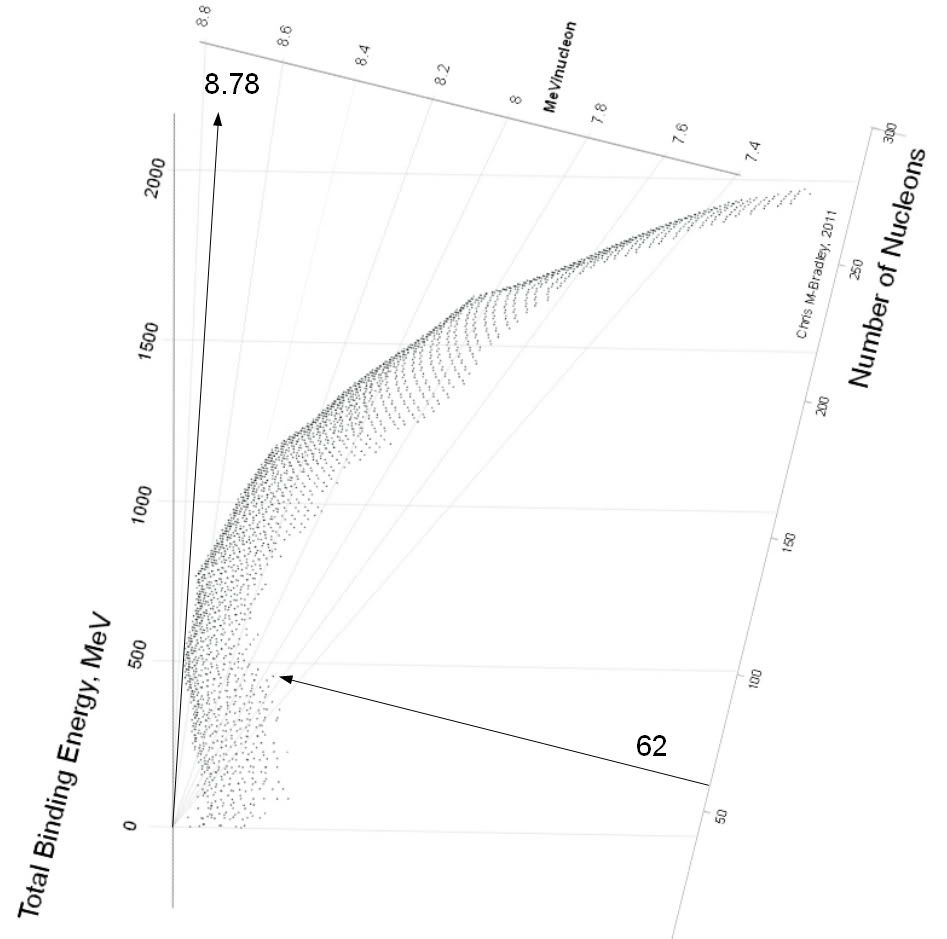
Total binding energy for 62Ni = 545.25881 MeVD Tibbets wrote:Solving for the equation for Ni62 yields:
energy yield for Ni62 + P ---> Cu63
8.9 MeV / nuclon * 62 nucleons ---> 8.7 MeV/ nucleon * 63 = 551 MeV ---> 548 MeV . The total binding energy drops by ~ 3 MeV. As this binding energy represents the energy released, it is clear that it took more energy to make the Ni62, than it did to make the Cu63. This is an endothermic reaction.
This would be correct if the values were real.D Tibbets wrote:Let me try some algebra.
...
Solving for the equation for Ni62 yields:
energy yield for Ni62 + P ---> Cu63
8.9 MeV / nuclon * 62 nucleons ---> 8.7 MeV/ nucleon * 63 = 551 MeV ---> 548 MeV . The total binding energy drops by ~ 3 MeV. As this binding energy represents the energy released, it is clear that it took more energy to make the Ni62, than it did to make the Cu63. This is an endothermic reaction.
Real numbers are ~ 8.78 for 62Ni and 8.758 for Cu63. Thus, using your arithmatic...Then he wrote: I used made up numbers or recollected ~ numbers for the binding energy per nucleon. If you feel that my results are biased, I invite you to use accurate numbers to do the calculations. If you find contrary results I will be flabbergasted.
Code: Select all
Ni: 8.78*62 ~ 544
Cu: 8.76*63 ~ 552My prevous post was as clear as mud, and also innacurate as well. I used faulty recall and numbers necessary to get the answer I wanted. In effect I am flabbergasted! My target was backwards (+/_ ) and my made up numbers were wrong.chrismb wrote:Total binding energy for 62Ni = 545.25881 MeVD Tibbets wrote:Solving for the equation for Ni62 yields:
energy yield for Ni62 + P ---> Cu63
8.9 MeV / nuclon * 62 nucleons ---> 8.7 MeV/ nucleon * 63 = 551 MeV ---> 548 MeV . The total binding energy drops by ~ 3 MeV. As this binding energy represents the energy released, it is clear that it took more energy to make the Ni62, than it did to make the Cu63. This is an endothermic reaction.
Total binding energy for 63Cu = 551.38125 MeV
I don't understand where you got your figures from, Dan.
------------------------------------------"The importance of knowing the nuclear binding energy per nucleon is that it tells us whether energy will be released in a particular nuclear reaction. If the somewhat weakly bound uranium nucleus (7.41 MeV/ nucleon) splits into two more tightly bound nuclei like cesium (8.16 MeV/nucleon) and zirconium (8.41 MeV/ nucleon), energy is released. At the other end of the graph, if we combine two weakly bound deuterium nuclei (2.8 MeV/nucleon) to form a more tightly bound Helium 4 nucleus (7.1 MeV/nucleon), energy is also released. Any reaction that moves us toward the Iron 56 nucleus releases energy. "
Which direction is your search going now, Dan. You've argued your case, it is OK to recognise the error.D Tibbets wrote:This and the additional links and quotes brings me closer to the truth. At least my truth
Dan, CHECK YOUR NUMBERS!!!D Tibbets wrote: "Ni62 = BE avg/nucleon 8.795 BE 545.259
Pb206 = BE avg/nucleon 7.875 BE 1622.693
Ca40 = BE avg/nucleon 8.551 BE 342.052"
.....
Cu63 = BE avg/nucleon= 8.76 , and the derived BE= 551.8 MeV
BE= BEave/nucleon * number of nucleons.
Examples based on the above numbers:
To add one nucleon to Ni62 = 8.795 BE avg/nucleon * 62 nucleons= 545.25 MeV (matches the number above)
Resultant Cu63= 8.76/ nucleon * 63 nucleons = 551 MeV.
The binding energy has increased by ~6 MeV
To add one nucleon to Ca40= 8.551/ nucleon *41 nucleons= 538 MeV. Compared to Ca 40 which has a BE of 342.052 MeV, this represents a BE of ~ - 196. The binding energy has decreased.
All of the cases result in increased binding energy. Your conclusion is incorrect.Then he wrote: To add one nucleon to Pb206 = 8.551/ nucleon *207 nucleons= 1630 MeV. Compared to Pb206 the binding energy has increased by ~ 7.5 MeV
Using these referenced and hopefully accurate numbers within these three example, several things are obvious. A fusion below Ni62 resulted in a loss of binding energy, while the two examples of of fusion above Ni62 resulted in increased binding energy.
Sigh, your right, I made a math error. The Ca40 --->Ca41 would be ~ 351 MeV total binding energy.KitemanSA wrote:Dan, CHECK YOUR NUMBERS!!!D Tibbets wrote: "Ni62 = BE avg/nucleon 8.795 BE 545.259
Pb206 = BE avg/nucleon 7.875 BE 1622.693
Ca40 = BE avg/nucleon 8.551 BE 342.052"
.....
Cu63 = BE avg/nucleon= 8.76 , and the derived BE= 551.8 MeV
BE= BEave/nucleon * number of nucleons.
Examples based on the above numbers:
To add one nucleon to Ni62 = 8.795 BE avg/nucleon * 62 nucleons= 545.25 MeV (matches the number above)
Resultant Cu63= 8.76/ nucleon * 63 nucleons = 551 MeV.
The binding energy has increased by ~6 MeV
To add one nucleon to Ca40= 8.551/ nucleon *41 nucleons= 538 MeV. Compared to Ca 40 which has a BE of 342.052 MeV, this represents a BE of ~ - 196. The binding energy has decreased.
Ca40= 8.551/ nucleon *41 nucleons= 351 not what you said. Binding energy INCREASES by ~ 9MeV!All of the cases result in increased binding energy. Your conclusion is incorrect.Then he wrote: To add one nucleon to Pb206 = 8.551/ nucleon *207 nucleons= 1630 MeV. Compared to Pb206 the binding energy has increased by ~ 7.5 MeV
Using these referenced and hopefully accurate numbers within these three example, several things are obvious. A fusion below Ni62 resulted in a loss of binding energy, while the two examples of of fusion above Ni62 resulted in increased binding energy.
Moron. Using 1 sig fig in a calculation of the change in the 6the and 7th decimal places of mass-energy is going to give you nonsense.D Tibbets wrote: Note that there are errors in this post. See my next post for a more reasoned and fact supported analysis.
Let me try some algebra.
Binding energy/ nucleon chart gives values from ~ 0- 8.9 MeV on the low side of Ni62 and ~ 8.9 MeV to ~ 6 MeV on the high side (these numbers are approximations).
Like I said, Moron. Proper way: c^2*(mass in-mass out) = energy change.D Tibbets wrote:
f(x) = a/b *c
f(x)= energy yield
a= binding energy
b= nucleon
c= number of nucleons in the nucleus
The values are emperically determined from the graph and I have linked at least one source derived ~ values.
Solving for the equation for Ni62 yields:
energy yield for Ni62 + P ---> Cu63
8.9 MeV / nuclon * 62 nucleons ---> 8.7 MeV/ nucleon * 63 = 551 MeV ---> 548 MeV .
Garbage in, Garbage out. The only trend you show is that you have no clue.D Tibbets wrote: The total binding energy drops by ~ 3 MeV. As this binding energy represents the energy released, it is clear that it took more energy to make the Ni62, than it did to make the Cu63. This is an endothermic reaction.
This is only one simple example, and admittedly the empirical MeV numbers per nucleon is estimates, but I think this shows the trend.
Sorry. I could work this sort of mass defect calculation out when I was a kid, it is VERY basic math (although deriving it and proving it is something I could not do until Calc III). So, yes, "Moron" is the term for someone who cannot manage to do the math - and yet publicly exposes their ignorance despite that. No one told Dan he had to defend Iron as the Most Stable Element to the point of absurdity.KitemanSA wrote:Wiz,
Thank you for your support, but please not that way. Play nice please?
Dan, for some reason, seems to be generally reasonable about all issues... except this one. He seems most willing to cook the books to get the answer he seems fixated on. Oh well.